About oncogenetic drivers
NTRK is emerging as an actionable biomarker and oncogenic driver across a wide range of tumour types1–9
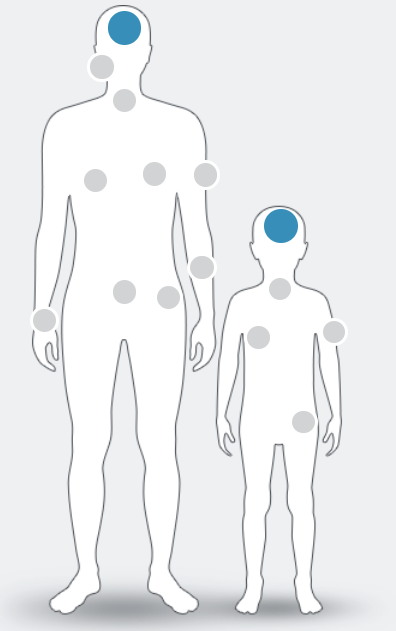
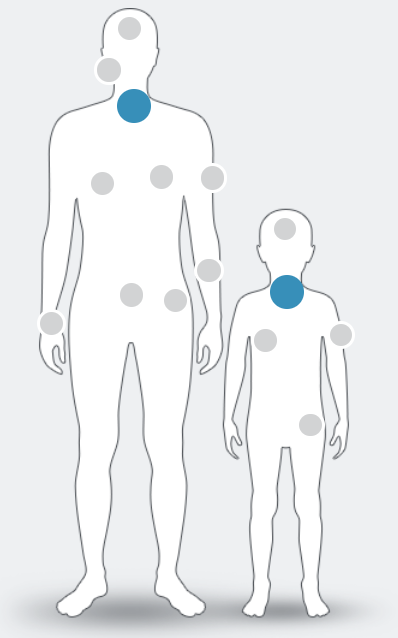
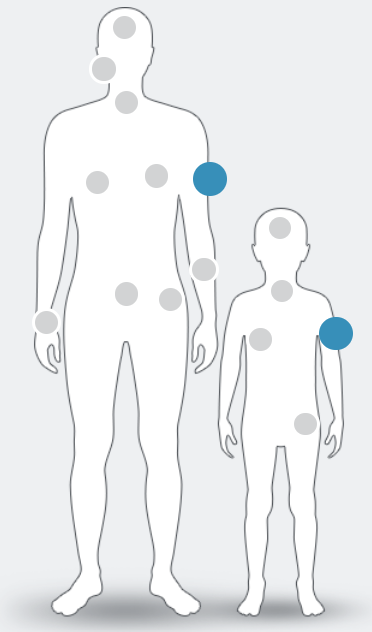
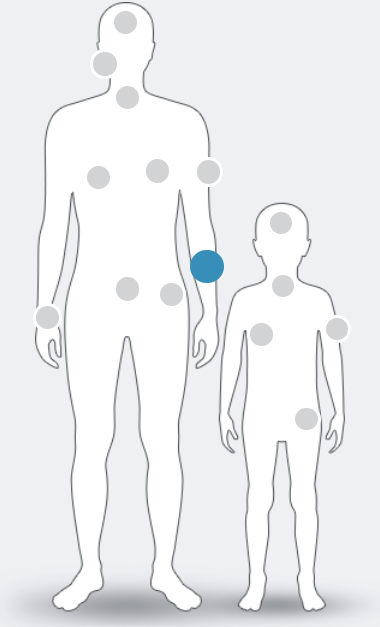
NTRK fusions have been identified in at least
25 tumour types in adult and paediatric patients, including:1–9
NTRK fusion+ cancer currently has no known defining clinical or pathological features. Only high-quality molecular testing such as next-generation sequencing (NGS) can confirm its presence1
ESMO proposal: NTRK testing10
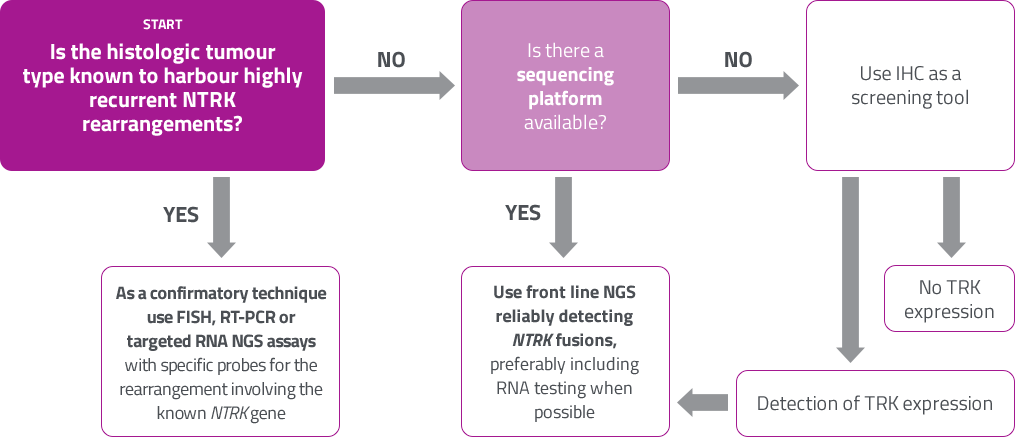
It is important to ensure that the diagnostic test covers NTRK 1, 2, 3 fusion genes and is validated with appropriate reference standards.
Figure adapted from Marchio C. et al , 2019.
High-quality molecular testing is needed to uncover NTRK fusion+ cancer1,11,12
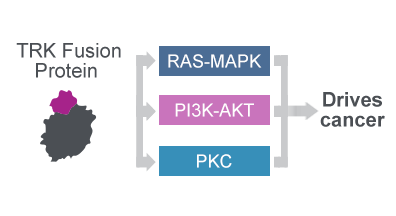
TRK fusion proteins drive cancer through aberrant signalling1,3-6
TRKs play an important role in healthy tissue
- The NTRK (neurotrophic tropomyosin receptor kinase) receptor family is encoded by the three NTRK genes that code for three proteins1
- In healthy tissue, the NTRK pathway is involved in the development and functioning of the nervous system as well as cell survival3,13

NTRK gene fusions create oncogenic proteins3
- Each of the three NTRK genes can combine with multiple other gene partners to create oncogenic fusion proteins1–3,14
- So far, 25 distinct fusions have been identified1

The oncogenic fusion proteins drive cancer through aberrant signalling1,3–6
- The oncogenic fusion proteins constitutively activate a signalling cascade implicated in cell proliferation, survival and angiogenesis1,3–6
Footnotes:
ESMO, European Society for Medical Oncology; FISH, DNA fluorescence in situ hybridisation; IHC, immunohistochemistry; NGS, next-generation sequencing; RT-PCR, reverse-transcriptase polymerase chain reaction.
1. Vaishnavi A, Le AT, Doebele RC. Cancer Discov 2015;5:25–34.
2. Lange AM, Lo HW. Cancers (Basel) 2018;10.
3. Amatu A, Sartore-Bianchi A, Siena S. ESMO Open 2016;1:e000023.
4. Khotskaya YB, et al. Pharmacol Ther 2017;173:58–66.
5. de Lartigue J. TRK inhibitors advance rapidly in “tumor-agnostic” paradigm. OncologyLive 2017;18. Available at: https://www.onclive.com/publications/oncology-live/2017/vol-18-no-15/trk-inhibitors-advance-rapidly-in-tumoragnostic-paradigm (Accessed July 2020).
6. Robbins HL, Hague A. Front Endocrinol (Lausanne) 2016;6:1–22.
7. Dupain C, et al. Mol Ther Nucleic Acids 2017;6:315–326.
8. Kummar S, Lassen UN. Target Oncol 2018;13:545–556.
9. Cocco E, Scaltriti M, Drilon A. Nat Rev Clin Oncol 2018;15:731–747.
10. Marchio C. et al. Ann Oncol 2019;30:1417–1427.
11. Murphy DA, et al. Appl Immunohisochem Mol Morphol 2017;25:513–523.
12. Rogers TM, et al. Sci Rep 2017;7:1–8.
13. Chong CR, et al. Clin Cancer Res 2017;23:204–213.
14. Stransky N, et al. Nat Commun 2014;5:4846.