
About oncogenetic drivers
ROS1 is a therapeutic target in NSCLC, with gene fusions occurring in 1–2%
of patients.1,2
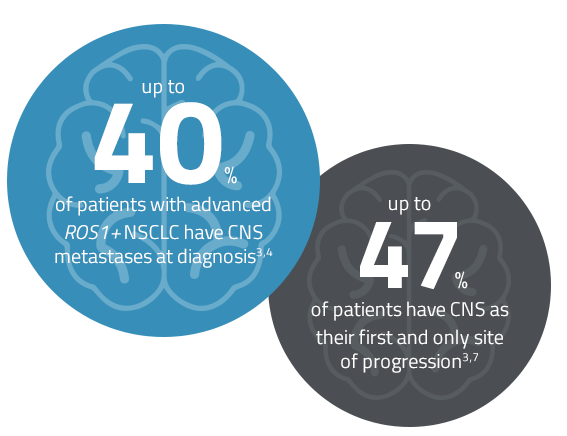
Patients with ROS1+ NSCLC require an effective treatment with both systemic and central nervous system (CNS) activity.3–6
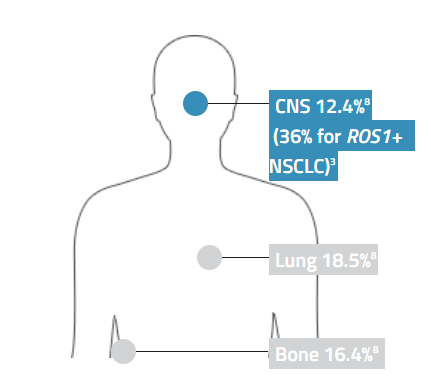
The most common sites of metastasis for patients with all types of stage IV lung cancer8
Despite advances in therapy for ROS1+ NSCLC, additional treatment options are needed to improve clinical outcomes3
High-quality molecular testing, such as FISH and NGS, is needed to confirm patients with actionable ROS1 gene fusions9
Testing for ROS1+ rearrangement should be systematically carried out in advanced NSCLC10
- ROS1 testing involves the detection of ROS1 gene rearrangements or overexpression of ROS1-fusion proteins11
- FISH, IHC and RT-PCR are currently used in routine clinical practice:

NGS is increasingly being used and is a sensitive and accurate test to identify most ROS1 gene fusions with a single test9,13–19
- NGS is emerging as a technology with the sensitivity and accuracy necessary to identify all ROS1 gene fusions with a single test9,13–19
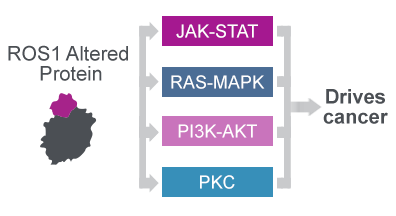
The ROS1 protein plays an important role in healthy tissue and if altered by fusion events or mutations drives cancer through aberrant signalling20-22
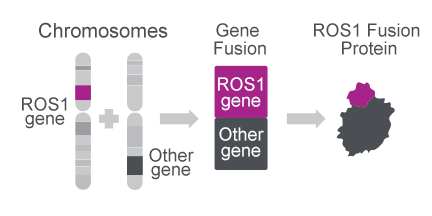
ROS1 gene fusions create oncogenic proteins22
- The ROS1 gene can combine with multiple other gene partners to create an oncogenic fusion protein22
- So far ~55 gene fusions have been detected28
The oncogenic ROS1 protein drives cancer through aberrant signalling22
- The oncogenic fusion or mutated ROS1 protein constitutively activates signalling cascades implicated in cell proliferation, survival and angiogenesis22
Footnotes:
CNS, central nervous system; FISH, fluorescence in situ hybridisation; IHC, immunohistochemistry; NGS, next-generation sequencing; NSCLC, non-small cell lung cancer; RT-PCR, reverse transcription polymerase chain reaction.
1. Bergethon K, et al. J Clin Oncol 2012;30:863–870.
2. Dugay F, et al. Oncotarget 2017;8:53336–53351.
3. Patil T, et al. J Thorac Oncol 2018;13:1717–1726.
4. Gainor JF, et al. JCO Precis Oncol 2017. DOI: 10.1200/PO.17.00063.
5. Mazières J, et al. J Clin Oncol 2015;33:992–999.
6. Wu YL, et al. J Clin Oncol 2018;36:1405–1411.
7. Xu H, et al. Cancer Med 2020;9:3328–3336.
8. Oikawa A, et al. Oncol Lett 2012;3:629–634.
9. Bubendorf L, et al. Virchows Arch 2016;469:489–503.
10. Planchard D. Ann Oncol 2018;29:iv192–iv237.
11. International Association for the Study of Lung Cancer. IASLC Atlas of ALK and ROS1 testing in lung cancer. Available at: https://www.iaslc.org/research-education/publications-resources-guidelines/iaslc-atlas-alk-and-ros1-testing-lung-cancer (Accessed November 2020).
12. Rossi G, et al. Lung Cancer (Auckl) 2017:8:45–55.
13. National Comprehensive Cancer Network. NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines). Non-Small Cell Lung Cancer. V.6.2020, 2020. Available at: https://www.nccn.org/professionals/physician_gls/pdf/nscl.pdf (Accessed November 2020).
14. Diaz L, Bardelli A. J Clin Oncol 2014;32:579–586.
15. Shan L, et al. PLoS One 2015;10:e0120422.
16. Cao B, et al. Onco Targets Ther 2016;31:131–138.
17. Zheng Z, et al. Nat Med 2014;20:1479–1484.
18. Drilon A, et al. Clin Cancer Res 2015;21:3631–3639.
19. Grada A, Weinbrecht K. J Invest Dermatol 2013;133:e11.
20. Birchmeier C, et al. Proc Natl Acad Sci U S A 1987;84: 9270–9274.
21. Rikova K, et al. Cell 2007;131:1190–1203.
22. Gainor J, Shaw A. Oncologist 2013;18:865–875.
28. Drillon et al. Clinical Oncology 2021; 18:35-55
