
Welcome to ACTRIMS-ECTRIMS 2020

An overview of the sessions and attendees at MSVirtual2020
The 8th Joint Annual Meeting of the Americans Committee for Treatment and Research in Multiple Sclerosis (ACTRIMS) and the European Committee for Treatment and Research in Multiple Sclerosis (ECTRIMS) took place under a new virtual format on 11–13 September 2020. Opening the meeting, which is the largest international conference dedicated to multiple sclerosis (MS) research, ACTRIMS President Jeffrey Cohen highlighted that the main focus of MSVirtual2020 would be advances and breakthroughs made in MS causes and risk factors, diagnostic tools, treatment response biomarkers, technology, therapies and interventions. ECTRIMS President Bernhard Hemmer added that this year’s meeting would continue to bring together world-class neurologists, clinicians and researchers in the field of MS from around the world to present and discuss latest research and therapeutic advances.

“You will hear about cutting edge research into the cause, biology, measurement, sequelae and treatment of MS.”
Jeffrey Cohen, ACTRIMS President

“Although we cannot meet in person, I’m sure you will take advantage of the flexibility of the virtual format of this year’s meeting.”
Bernhard Hemmer, ECTRIMS President
Immunopathogenesis of MS
Stefan Bittner, University of Mainz, Mainz, Germany, provided an overview of new insights on the immunopathogenesis of MS. Dr Bittner highlighted that while disease-modifying treatments (DMTs) can reduce MS relapses, they are unable to prevent disease progression and patients subsequently develop secondary progressive MS (SPMS). Current pathophysiological models of progressive disease include:
- The involvement of the adaptive versus innate immune system
- Immune-independent neurodegenerative pathways
- Repair mechanisms and higher network alterations
Central nervous system (CNS)-intrinsic pathways, the complex interplay between immune cells, glial cells and neurons within the CNS parenchyma, also appears to impact on brain function both in health and disease. Neuroinflammation-induced network alterations result in cortical hyperexcitability and cell death, leading to neurodegeneration in the long term. Neuroinflammation appears to induce toxic protein accumulation in neurons, which requires rescue by pharmacological proteasome activation. Calcium influx via nanoscale membrane ruptures also appears to promote axonal degeneration in experimental autoimmune encephalomyelitis (EAE). Of note, beneficial CNS-immune crosstalk involving the fast-direct neuronal interleukin (IL)-4R signalling pathway has been reported. Independent from T cell function, IL-4 binds IL-4R on neurons, promoting axonal integrity and neuronal recovery in EAE. Newly recognised regulators of immune pathways and CNS-specific functions of T cells which may influence progression of MS include T helper (Th)17 cells and some aspects of nutrition (e.g. dietary tryptophan, short-chain fatty acid pentanoate and propionic acid). T cells directly target the grey matter in progressive MS, with β-synuclein suggested as a late-stage autoimmune target given that it is elevated in progressive MS (but not in relapsing/remitting MS [RRMS]).
“There is an ongoing need for new avenues of research that address the problem of MS progression in a fundamentally different way.”
Stefan Bittner, University of Mainz, Mainz, Germany
Diagnosis
Helen Tremlett, University of British Columbia, Canada, summarised and reflected on what is currently known, and not known, about the MS prodrome. A prodrome can be defined as an early set of signs or symptoms occurring before the classical onset of a disease. Dr Tremlett highlighted that the MS prodrome had largely been dismissed in the early literature, in contrast with prodromal phases already being recognised and well accepted for other neurological diseases, such as Parkinson’s disease. However, the MS prodrome has started to receive greater consideration in recent years. For a chronic disease such as MS, a prodromal phase could span months or even years prior to the onset of typical MS symptoms. Thus, a better understanding of the MS prodrome could have profound implications for disease prevention and management. While an understanding of the MS prodrome continues to develop, Dr Tremlett noted that there is sufficient knowledge of the MS prodrome to affect current practice now, particularly research into risk factors for MS onset, although more work is required before it can have direct application on routine practice. A myriad of putative risk factors and features of the MS prodrome – measurable via healthcare use – require careful evaluation, although no single feature appears specific to MS. For example, frequent use of healthcare in patients with MS (vs controls) in the 5 years prior to a first demyelinating event may suggest the existence of a measurable MS prodrome. These findings have clinical and research implications, including the establishment of an earlier window of opportunity to identify and potentially treat MS – integrating these symptoms in the diagnostic procedure may help earlier disease identification.
“The prodrome remains poorly understood but needs to be considered very carefully when searching for risk factors for MS onset.”
Helen Tremlett, University of British Columbia, Canada
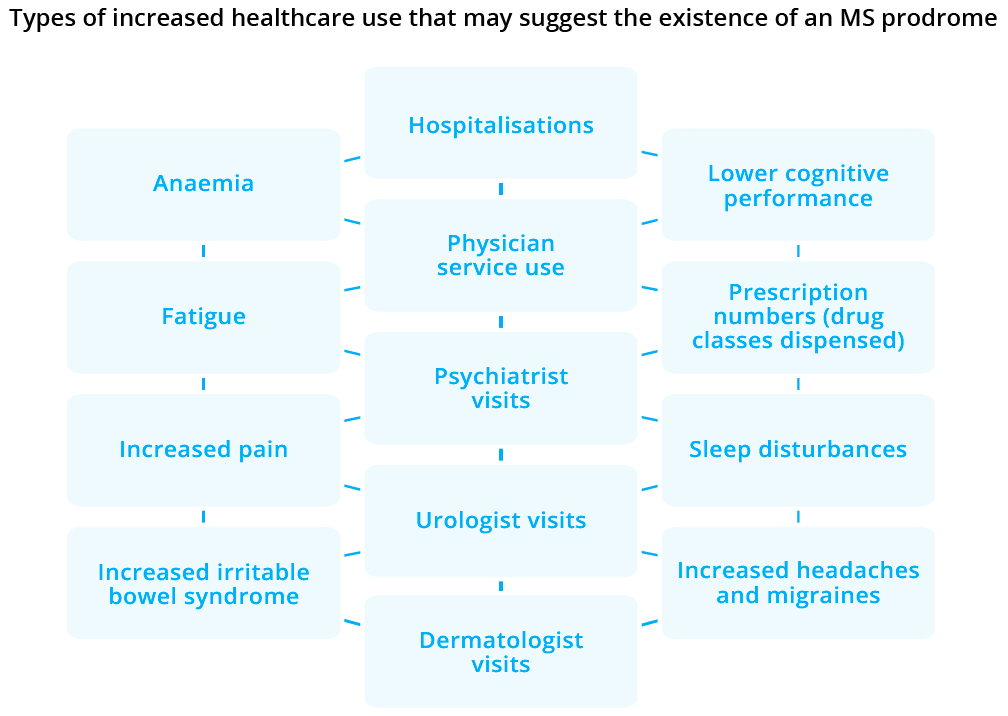
A myriad of putative risk factors and features of the MS prodrome are measurable via healthcare use
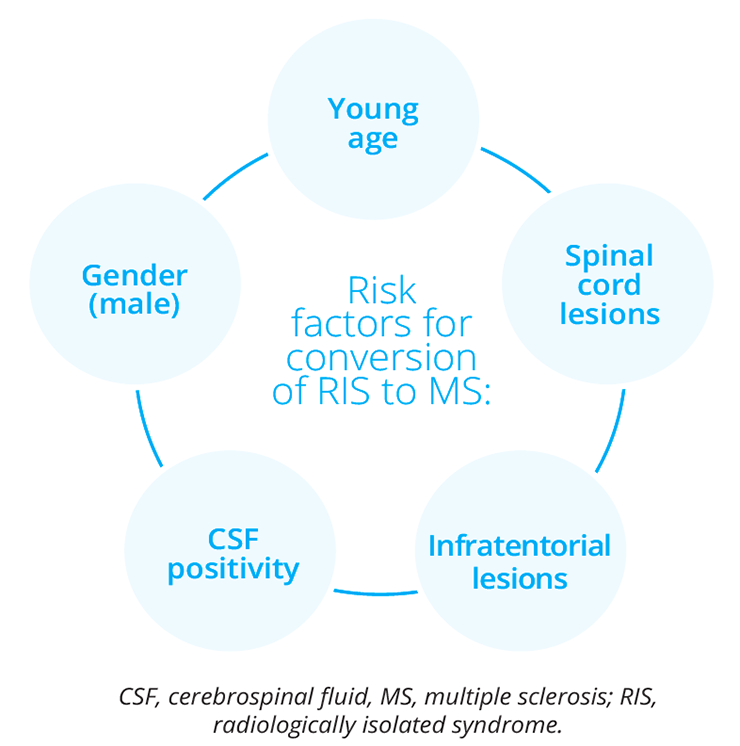
Risk factors for conversion of RIS to MS
Christine Lebrun-Frénay, Centre Hospitalier University de Nice, France, provided a brief overview of the diagnosis and management of radiologically isolated syndrome (RIS), a recently described MS subtype that expands upon the phenotype of at-risk individuals for future demyelinating events. Healthy individuals who do not exhibit signs of neurological dysfunction commonly have brain magnetic resonance imaging (MRI) studies. These studies are performed for a reason other than an evaluation for MS, revealing unexpected anomalies highly suggestive of demyelinating plaques given their size, location and morphology. These healthy individuals lack symptomatology suggestive of MS and fulfil formal criteria for RIS. The presence of asymptomatic lesions in the cervical cord provides an increased risk of progression, either to relapsing or progressive MS. Further elucidation of the natural disease course of RIS is required, particularly in asymptomatic patients.
“Up to 30–45% of patients presenting with a RIS will present with clinical progression.”
Christine Lebrun-Frénay, Centre Hospitalier University de Nice, France
Treatment of MS
Mar Tintoré, Hospital Universitari Vall d’Hebron, Barcelona, Spain, explored appropriate MS disease therapy for individual patients. Dr Tintoré noted that from 1994 to 2017, there has been an 84% reduction in the mean time from clinically isolated syndrome to the initiation of DMTs. In addition, during the past decade the entire course of MS from the first sign of a demyelinating disorder through the progressive course appears to be milder due to a complex interplay of several factors, such as:
- Diagnostic criteria
- Changes in the epidemiology of MS
- Impact of early and appropriate DMT
- Improvement of the general state of health in the population
However, the natural history of MS is highly heterogeneous and a subgroup of patients has been identified with what might be termed ‘aggressive MS’. These patients may have frequent, severe relapses with incomplete recovery and are at risk of developing greater and permanent disability at the earlier stages of the disease. The therapeutic window of opportunity for these patients appears narrow, and while they could benefit from early treatment with a highly efficacious treatment, a unified definition of aggressive MS does not exist and available data on its treatment remain sparse. A number of prognostic factors inform individual treatment strategies.
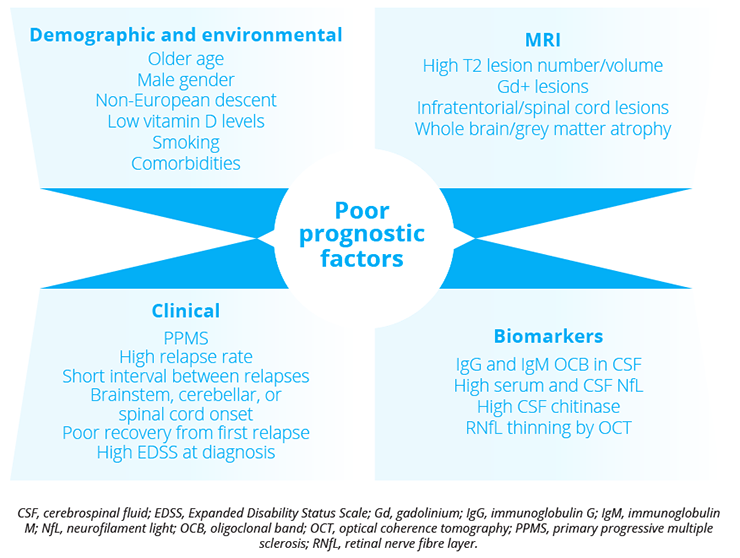
Poor prognostic factors for MS treatment include patient demographics, environmental influences, MRI, clinical aspects and biomarkers.
In addition, a number of patient and geographical factors should also influence individual treatment strategies:
- Comorbidities
- Access to reimbursement
- Reproductive considerations
- Risk tolerance
- Acceptability of route and schedule of administration
- Perceived burden
“Comorbidities such as epilepsy, migraine and hyperlipidaemia are associated with increased risk of relapse and disability.”
Mar Tintoré, Hospital Universitari Vall d’Hebron, Barcelona, Spain
Ellen Mowry, Johns Hopkins University School of Medicine, Baltimore, USA, provided a brief overview of escalation versus early aggressive treatment algorithms. Dr Mowry explained that with the availability of nearly 20 medications approved for relapsing forms of MS, there is growing pressure to better define how to select the most appropriate treatment for a given individual, particularly at the time of diagnosis. When few therapies were available, the typical treatment approach was of ‘escalation’. However, with the advent of more therapies, the treatment strategy has become less clear. Some opinion pieces have called for the use of higher-efficacy DMTs at an early/first-line stage, although a lack of definitive data has led to considerable debate amongst physicians. Randomised clinical trials (RCTs) have demonstrated short-term benefits of some specific higher-efficacy therapies with respect to disability outcomes compared with another, specific moderate-efficacy therapy, but these trials have not tested treatment strategies; in particular, escalation after ongoing MS activity is not typically allowed in such protocols. The current ‘push’ for aggressive first treatment for all patients is not evidence-based, with observational and randomised escalation studies also having limitations, such as a lack of clarity if treatments have been escalated in a timely manner when breakthrough disease occurred. In addition, payers still deny or restrict formulary with respect to higher-efficacy therapies, demanding RCT evidence. Patients also remain cautious of the possible risks associated early aggressive therapy, such as infections, with real-world evidence unable to monitor and mitigate potential risk to the same extent as RCTs. Two ongoing randomised studies – TRaditional versus Early Aggressive Therapy for MS (TREAT-MS; NCT03500328) and Determining the Effectiveness of earLy Intensive Versus Escalation Approaches for RRMS (DELIVER-MS; NCT03535298) – hope to identify if specific treatment strategies in the relapsing-remitting phase of MS can prevent, delay or reduce intermediate- to longer-term disability accrual.
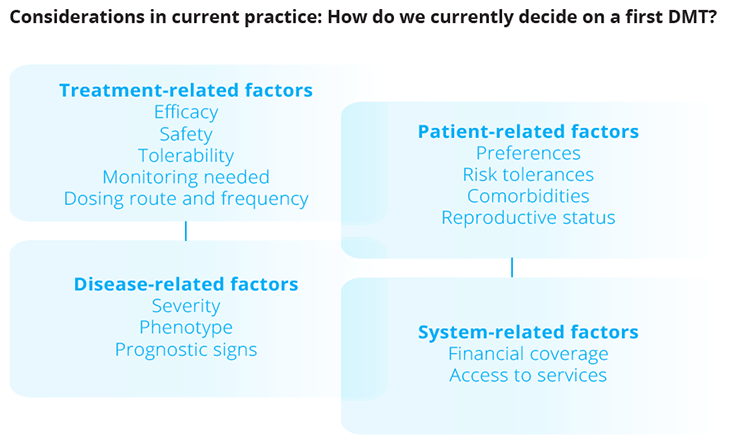
Factors that influence DMT use.
“Patients, physicians and payers need more definitive data to support early aggressive treatment choices in relapsing MS.”
Ellen Mowry, Johns Hopkins University School of Medicine, Baltimore, USA
Gavin Giovannoni, Barts and The London School of Medicine and Dentistry, UK, discussed the customised management of patients with MS using approved therapies. Professor Giovannoni explained that the overall aim was to encourage the widespread adoption of a therapeutic strategy that aimed to maximise the lifelong brain health of every individual with MS. The course of MS has been improved by earlier diagnosis and the increased availability of DMTs. However, apart from anti-drug or neutralising antibodies, there are no validated baseline predictors of who will be responders or non-responders to DMTs. High-efficacy therapy commenced within 2 years of disease onset is associated with less disability after 6–10 years than when commenced later in the disease course. This finding can inform decisions regarding optimal sequence and timing of MS therapy. However, customised medicine is not only about efficacy – a multitude of other factors may also require careful consideration.
“More efficacious DMTs simply increase the overall chances of a patient with MS being a responder.”
Gavin Giovannoni, Barts and The London School of Medicine and Dentistry, London, UK
Given that there is currently no consensus on when and how aggressively to treat patients with RRMS, Pietro Iaffaldano, University of Bari “Aldo Moro”, Bari, Italy, presented Italian Registry MS data which suggested that an early intensive treatment strategy was more effective than an escalation treatment strategy in controlling disability progression in patients with relapsing MS. Of note, the treatment effect tended to increase over time despite patients in the escalation treatment strategy group being escalated to a higher-efficacy DMT. This may be due to a more potent effect on disability progression of the high-efficacy DMTs in the early years of the disease or, alternatively, to the longer exposure to them.
“These findings may drive treatment decisions of physicians, particularly in the cases of naïve patients with poor prognostic factors at the onset of disease.”
Pietro Iaffaldano, University of Bari “Aldo Moro”, Bari, Italy
Therapeutic research in MS has typically focused on the development of treatments with little investigation regarding the possibility of discontinuation of DMTs. One aspect of DMT discontinuation is to limit the impact on the so-called ‘cortical morbidities’. John Corboy, University of Colorado, Denver, USA, briefly explored the appropriateness of discontinuing DMT in patients with MS. The question to ask is “Is there an ongoing benefit of using the medication and does that benefit outweigh potential risk?”. As a patient ages, if the benefit of a given DMT reduces while the risk increases, a time may come when the treatment benefit may stop outweighing the risk; for expensive MS medications such as DMTs, there may be a strong clinical opinion to discontinue the DMT. However, approximately two-thirds of patients (66.3% of 377 responders in a US-based MS survey) would be unwilling to stop taking DMTs, regardless of physicians’ opinions, even if their disease was considered to be deemed ‘non-active’; this may be due to potential rapid disease progression or a rebound of inflammatory disease activity. The ongoing Discontinuation of DMTs in MS (DISCOMS; NCT03073603) study is specifically looking at outcomes of treatment discontinuation in patients aged >55 years, such as inflammatory disease activity, relapse within 5 years, and lesions on MRI within 3 years of discontinuation. The study is scheduled to end in 2021.
“There is a lack of knowledge on when and why to stop using DMTs with MS patients.”
John Corboy, University of Colorado, Denver, USA
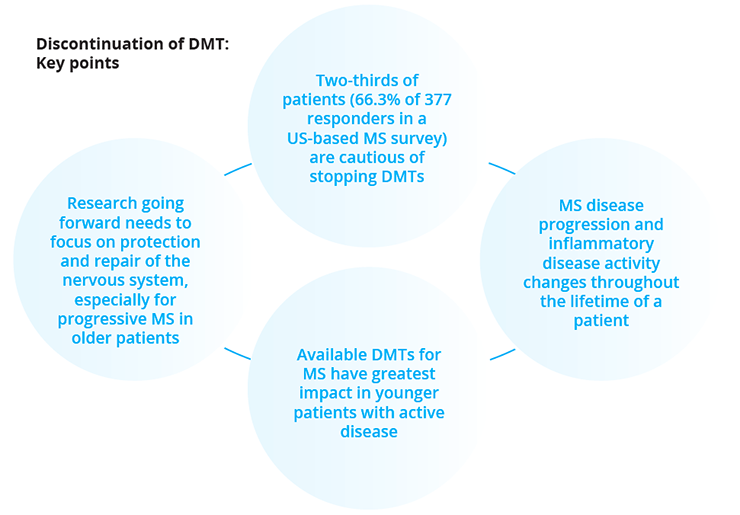
Key points when considering DMT discontinuation
Fredrik Piehl, Karolinska Institutet, Stockholm, Sweden, provided a brief overview of B-cell directed therapy in MS.
Lars Forsberg, Karolinska Institutet, Stockholm, Sweden, discussed the validation of three SPMS classification methods in five European MS registries within the SPMS Research Collaboration Network. Using data from these registries, the following three Expanded Disability Status Scale (EDSS)-centric classification methods were tested for overall classification performance based on sensitivity, specificity, and accuracy:
- Modified real-world EXPAND criteria
- Data-derived definition from University of Melbourne (without pyramidal Functional Score)
- Decision tree classifier from Karolinska Institutet
The findings from the study suggested that all three methods could be used to objectively assign SPMS with a fairly high performance for registry studies. Previous RCTs and real-world studies have generally failed to demonstrate therapeutic benefits of DMTs in patients with primary progressive MS (PPMS). Mattia Fonderico, University of Florence, Florence, Italy, used data from the Italian MS Registry to investigate the effectiveness of DMT on hard disability outcomes (EDSS 6 and 7) in a real-life population of patients with PPMS. Dr Fonderico demonstrated that longer exposure to DMTs may delay disability progression in this patient group. In addition, a subgroup analysis suggested that treating younger patients and earlier treatment initiation may improve the patients’ long-term disability outcomes.
“Future studies need to evaluate long-term safety of DMTs in order to maximise the benefit-risk balance, as well as to assess whether and when it is of no value to continue a DMT in PPMS.”
Mattia Fonderico, University of Florence, Florence, Italy
Biomarkers
Özgür Yaldizli, University Hospital Basel, Basel, Switzerland, reported on the value of serum neurofilament light (sNfL) chain levels as a biomarker of suboptimal treatment response in patients with MS in clinical practice. NfL in blood may have potential as a biomarker of neuroaxonal loss in MS. While numerous studies have shown that sNfL is associated with clinical and MRI disease activity and treatment response, it is not clear whether it, under established DMT, can identify patients with suboptimal treatment response. Using data from the real-world Swiss MS Cohort Study, Dr Yaldizli demonstrated that serum sNfL was independently associated with clinical and MRI measures of disease activity. Importantly, increased sNfL in DMT-treated patients with MS predicted relapse/EDSS worsening and MRI activity in the subsequent year. These data support the value of serum NfL for treatment monitoring in MS clinical practice.
“sNfL reflects neuro-axonal damage and may be suitable as a biomarker of suboptimal response to DMTs.”
Özgür Yaldizli, University Hospital Basel, Basel, Switzerland
Strategies to promote remyelination in MS
Professor Robin Franklin, Wellcome-Medical Research Council Cambridge Stem Cell Institute, Cambridge, UK, highlighted that two modes of remyelination have been identified:
- Remyelination by newly generated oligodendrocytes
- Remyelination by surviving oligodendrocytes
The efficiency of remyelination declines progressively with adult ageing. This is due, in part, to intrinsic changes within adult CNS progenitors. Experimental studies have revealed that the age effects are reversible. Targeting the biology of the ageing CNS progenitor offers a valuable approach to countering the transition from RRMS to SPMS. Professor Gianvito Martino, Vita Salute San Raffaele University, Milan, Italy, briefly explored a possibility for cellular therapy to enhance remyelination. Different types of stem cells may have a role in both remyelination and immunomodulation:
- Haematopoietic stem cells
- Mesenchymal stem cells
- Neural stem cells
Remyelination can be achieved by mobilising endogenous stem cells and by transplanting stem cells. However, regeneration of long tract axons and neurons by stem cell therapy remains difficult to achieve at the present time.
Professor Catherine Lubetzki, AP-HP Sorbonne University, Paris, France, highlighted that an increased understanding of the remyelination process is needed to allow the identification of potential therapeutic targets. While the remyelination strategy is actively developing within MS, there remains a need for larger studies, novel trial designs and markers of repair.
Astrocytes are known to play critical roles in CNS development, homeostasis and response to injury. In addition to well-defined functions in synaptic signalling and blood-brain barrier control, astrocytes are now emerging as important contributors to white matter health. Veronique Miron, University of Edinburgh, UK, reviewed the roles of astrocytes in myelin formation and regeneration (remyelination), focusing on both direct interactions with oligodendrocyte lineage cells, and indirect influences via crosstalk with CNS resident macrophages (microglia). Dr Miron highlighted that dual functions of astrocytes in remyelination are regulated by nuclear factor erythroid 2-related factor 2 (Nrf2), the transcriptional master regulator of the stress-induced cytoprotective response, with sustained Nrf2 in astrocytes reducing reactivity during remyelination.
“Nrf2 is a molecular switch regulating the capacity of astrocytes to support remyelination.”
Veronique Miron, University of Edinburgh, UK
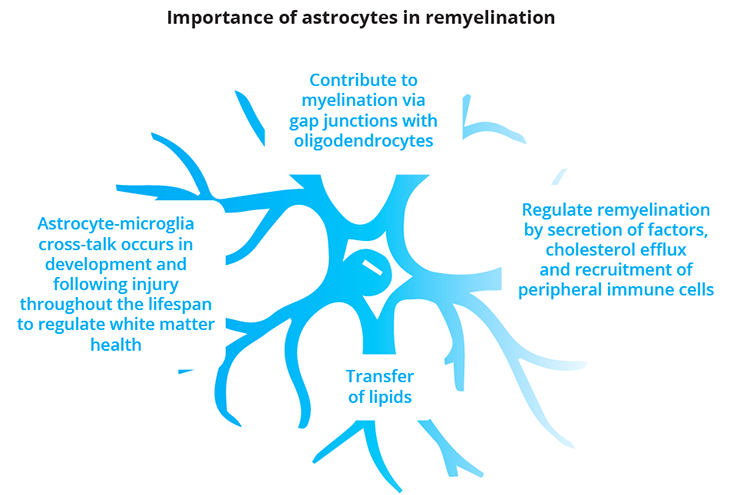
The multiple roles of astrocytes in remyelination
Jeffrey Huang, Georgetown University, Washington, USA, explored the role of amino acid transport in CNS inflammation and remyelination. Dr Huang presented data from a simple yet powerful method of labelling CNS demyelinating lesions by the intraperitoneal injection of a vital dye, neutral red, into mice to track and analyse changes that occur within the MS lesion microenvironment over time. These data included the finding that reduction of amino acid-fuelled inflammation through Slc7a5 inhibition improved CNS remyelination.
Management of MS-related symptoms

Fatigue is the most common, incapacitating symptom in patients with MS
Fatigue remains the most common symptom in patients with MS. Tiffany Joy Braley, University of Michigan, Ann Arbor, USA, provided an overview of rehabilitative and pharmacological therapies for MS-related fatigue. Fatigue affects up to 90% of patients with MS globally and is a strong predictor of:

Primary causes of MS-related fatigue include the influence of cytokines and direct CNS damage, while secondary physiological causes can include sleep disorders and chronic MS symptoms (e.g. pain and spasticity). Psychological factors such as depression/anxiety and poor resilience may further worsen fatigue. Cognitive behavioral therapy (CBT) can be used to target maladaptive thoughts and beliefs that can perpetuate symptoms of fatigue. However, patient access to CBT remains limited. Exercise-based strategies, which may promote neuroplasticity, reduce deconditioning and improve mood/sleep appear to have moderate positive effects on MS-related fatigue. Methylphenidate, modafinil and amantadine are commonly prescribed medications for alleviating fatigue in MS; however, none are approved for this indication by the FDA or EMA and available evidence supporting their efficacy remains both sparse and contradictory.
Rehabilitation and pharmacological approaches to MS-associated fatigue
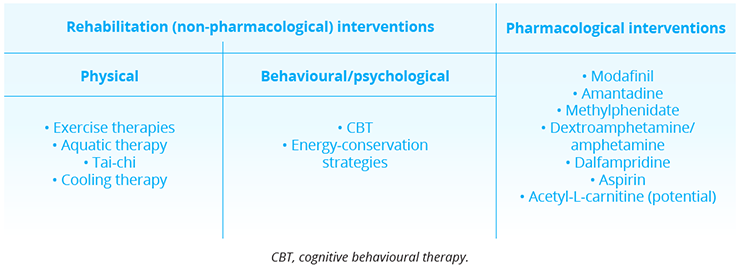
Rehabilitation and pharmacological interventions to MS-associated fatigue
“Fatigue is one of the most common and consequential symptoms of MS, yet it remains one of the most challenging symptoms to treat.”
Tiffany Joy Braley, University of Michigan, Ann Arbor, USA
Bardia Nourbakhsh, Johns Hopkins University, Baltimore, USA, presented data from a randomised crossover study (TRIUMPHANT-MS; NCT03185065) which compared the efficacy of methylphenidate, modafinil and amantadine (used twice daily for 6 weeks) against each other and placebo in patients with MS-related fatigue. The primary outcome measure was the Modified Fatigue Impact Scale measured while taking the highest tolerated dose. Methylphenidate, modafinil and amantadine did not significantly improve MS-related fatigue compared with placebo. In addition, higher proportions of participants reported adverse events while taking amantadine (38.6%), modafinil (40.0%), and methylphenidate (39.5%) as compared with placebo (30.6%).
“TRIUMPHANT-MS does not support the indiscriminate use of amantadine, modafinil and methylphenidate for treatment of fatigue in MS.”
Bardia Nourbakhsh, Johns Hopkins University, Baltimore, USA
Neuromyelitis optica spectrum disorders (NMOSDs)
NMOSD attacks tend to be more severe than MS attacks, with less recovery. The burden of NMOSD remains substantial, with relapse severity and frequency having an impact on disability and QoL.
Early and effective treatment to prevent relapse is key to the prevention of irreversible disability. Until recently, there were no approved DMTs for NMOSD, with off-label immunosuppressants azathioprine, mycophenolate mofetil and rituximab being most widely used. However, Phase III data with three new drugs have demonstrated a reduced risk of NMOSD attack/relapse:
- Eculizumab (monoclonal antibody against C5)
- Inebilizumab (monoclonal antibody against CD19)
- Satralizumab (monoclonal antibody against IL6)
“We now have valid diagnostic criteria and approved therapies to prevent relapses in NMOSD. ”
Ho Jin Kim, National Cancer Center, Korea
Closing Remarks
In response to the COVID-19 pandemic, the 8th Joint Annual Meeting of ACTRIM/ECTRIMS successfully took place under a new virtual format – MSVirtual2020. This virtual meeting maintained the aim of achieving an engaging, interactive and educational meeting experience for participants from around the world. Three days of cutting-edge research on the pathogenesis, diagnosis and treatment of MS and related disorders such as NMOSD were provided. Delegates were able to view the latest data, clinical experiences, patient perspectives and best practices that will hopefully stimulate new ways of thinking and ultimately translate into optimal patient care within the neurology field.
“We hope MSVirtual2020 inspired new ideas as well as dialogue and collaborative relationships between MS investigators around the globe.”
Benjamin Segal, Scientific Program Committee Co-Chair
©Springer Healthcare 2020. This content has been independently selected and developed by Springer Healthcare and licensed by Roche for Medically. The topics covered are based on therapeutic areas specified by Roche. This content is not intended for use by healthcare professionals in the UK, US or Australia. Inclusion or exclusion of any product does not imply its use is either advocated or rejected. Use of trade names is for product identification only and does not imply endorsement. Opinions expressed do not reflect the views of Springer Healthcare. Springer Healthcare assumes no responsibility for any injury or damage to persons or property arising out of, or related to, any use of the material or to any errors or omissions. Please consult the latest prescribing information from the manufacturer for any products mentioned in this material.